
The Modern Periodic Table Various Characteristics of Periods and Groups Group 1 is located on the left side of the periodic table, while group 18 is located on the right side. The elements in a given group do not have consecutive atomic numbers. The number of groupings varies from one to eighteen. In the long form of the periodic table, there are 18 groups. 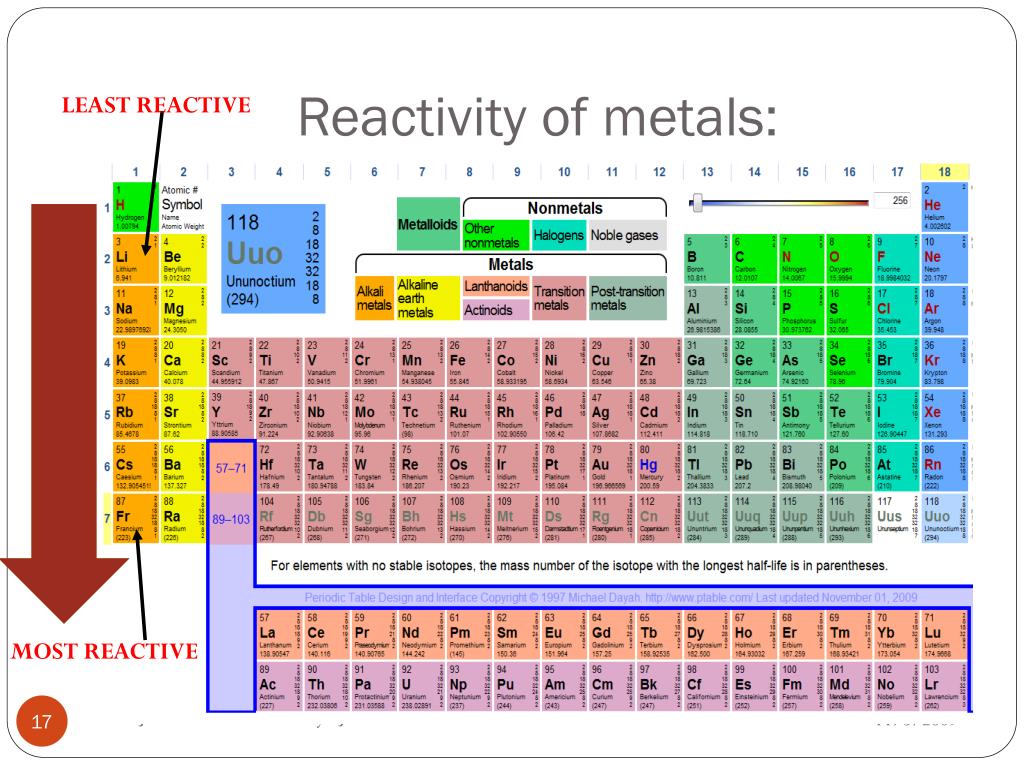
Groups are the vertical columns in a periodic table. The number of elements in a period is determined by the maximum number of electrons that may be accommodated in the various shells of an atom. The remaining elements are found in the seventh period. There are 32 elements in the sixth period.There are 18 elements in the fifth period.There are 18 elements in the fourth period.There are eight elements in the third period.There are eight elements in the second period. 
A “very short time” is how it’s described.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
